Details on additional Accord products mentioned can be found at the bottom of the page.
Role Of Testosterone
Testosterone is the most important hormone in male health, and is essential for regulating sex differentiation, producing male sex characteristics, spermatogenesis and fertility among other things.1,2
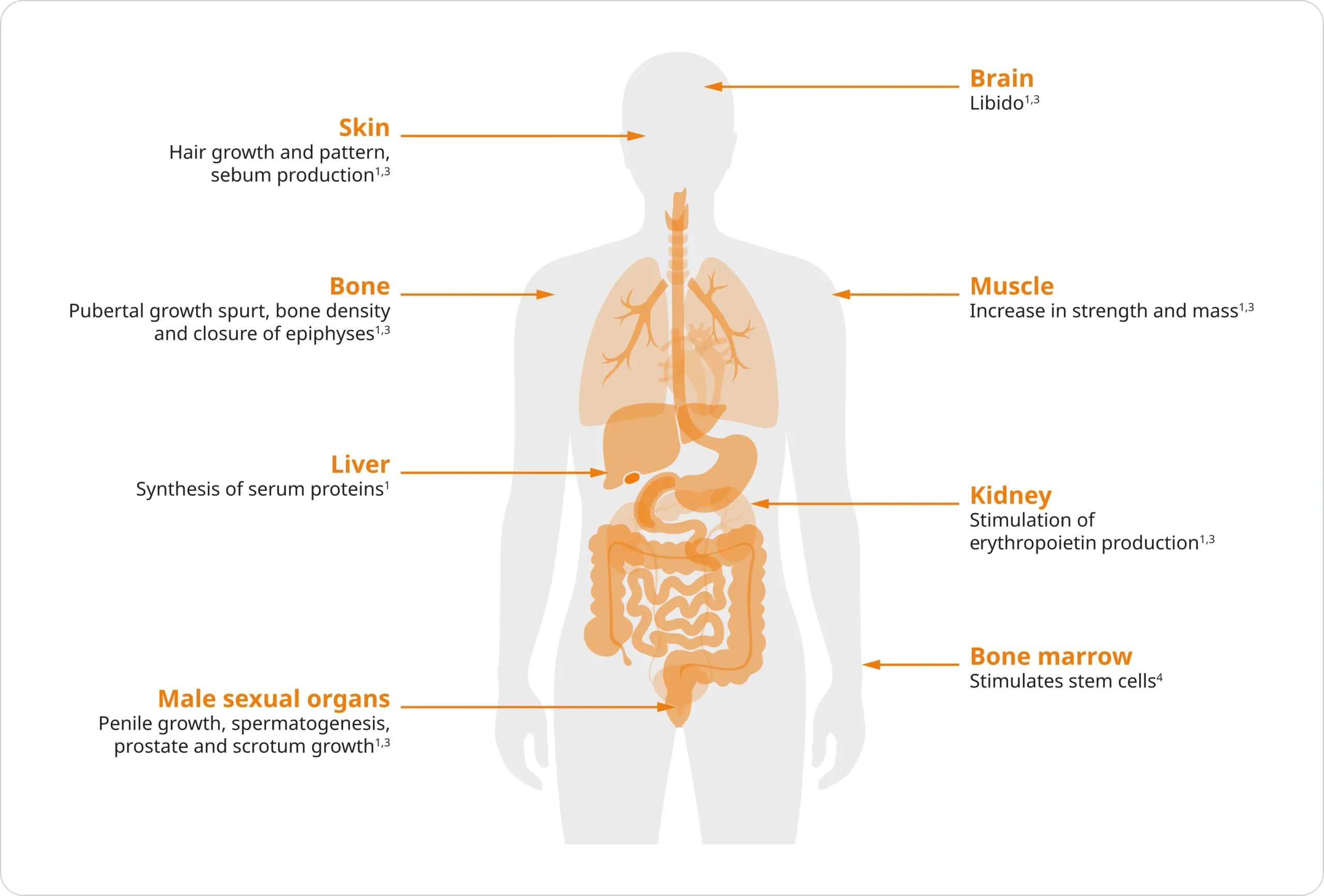
As men age, testosterone levels decrease. Testosterone deficiency may present with the following clinical signs and symptoms:1,2
- Sexual dysfunction (low libido and erectile dysfunction)
- Decreased skeletal muscle mass and strength
- Increased ratio of fat to lean body mass
- Decrease in bone mineral density and/or osteoporosis
- Decreased body hair
- Reduced mobility and energy
- Poor ability to concentrate
- Hot flushes
History Of Androgen Deprivation Therapy (ADT)
For more than 80 years, ADT has been the mainstay of advanced prostate cancer management.5,6
Treatments to suppress testosterone have expanded beyond surgical castration and oestrogen to include steroidal and non-steroidal antiandrogens, gonadotropin-releasing hormone (GnRH) agonists, and, most recently, GnRH antagonists.5
The development of such an extensive therapeutic armamentarium began with the pioneering work of Huggins and Hodges, who established testosterone as essential for prostate tumour growth.5,7
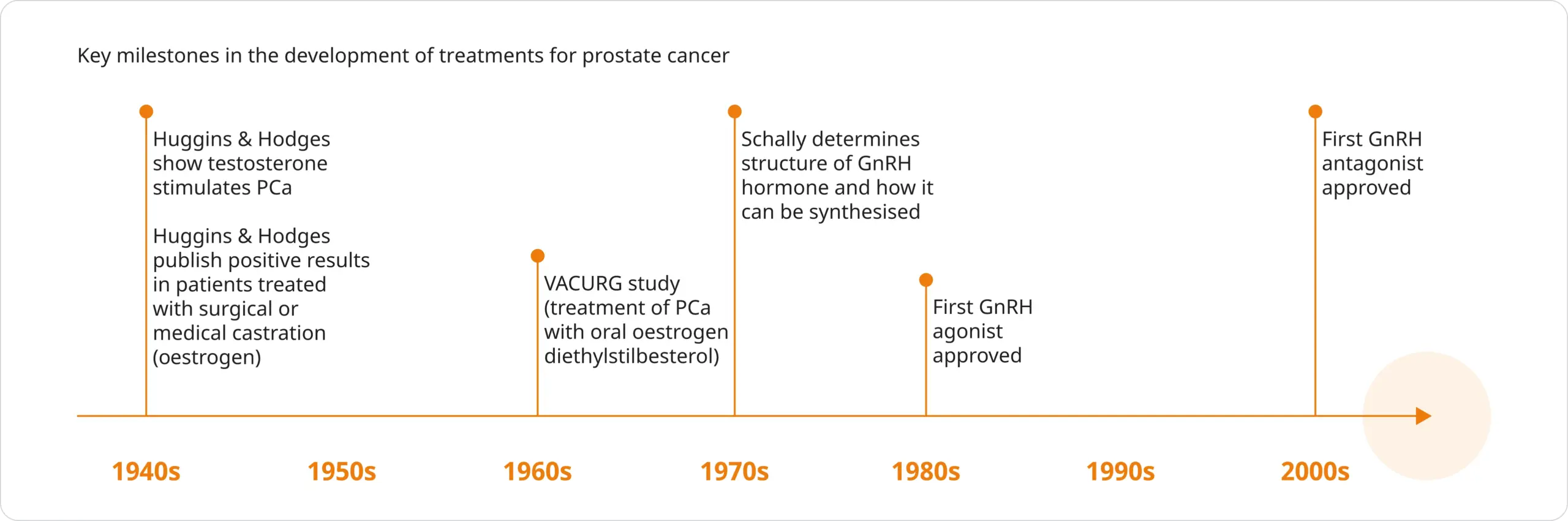
Adapted from Lehtonen & Kellokumpu-Lehtinen. 2023.7 & Drudge-Coates. 2010.8 & Denmeade & Isaacs. 2002.9 Abbreviations can be found in the footnotes of this page.
- The first major discovery was made in 1941 when Huggins and Hodges demonstrated that testosterone stimulates prostate cancer. They later published a paper describing the effects of treating advanced prostate cancer patients by surgical or medical castration via oral oestrogen administration7–9
- The Veterans Administration Cooperative Urologic Research Group (VACURG) study in the 1960s was one of the first large clinical trials assessing castration in men with prostate cancer. This study compared the effects of treating prostate cancer patients with oral oestrogen diethylstilbesterol. The study concluded oral oestrogen diethylstilbesterol was as effective as an orchiectomy in the treatment of prostate cancer9
- In 1971, Schally first isolated and identified GnRH and determined how it could be synthesised7–9
- The first GnRH agonists and antagonists were approved in the 1980s and 2000s, respectively7,8
Choosing An ADT Modality
As the mainstay treatment approach for advanced prostate cancer, ADT aims to reduce testosterone levels or block the peripheral action of testosterone through androgen blockade. This can be achieved with surgical castration or pharmacological agents.10,11
Several ADT modalities are available, differing in their mechanism of action, frequency and routes of administration.10,11
Orchiectomy
Surgical removal of the testicles remains a viable option to eliminate testicular production of testosterone. Performed under local anaesthesia, bilateral orchiectomy is the quickest way to achieve castration levels of testosterone (<50 ng/dL). However, it cannot be reversed and doesn’t allow for intermittent treatment.11,12
GnRH agonists
In clinical practice, GnRH agonists are the most commonly utilised forms of ADT and are available as intramuscular or subcutaneous injections on a 1-, 3-, 6-monthly or yearly basis.10–12
GnRH agonists bind to the GnRH receptor in the anterior pituitary gland, stimulating luteinising hormone (LH) and follicle-stimulating hormone (FSH) secretion. This, in turn, increases testosterone production, causing an initial surge.10
Increases in testosterone are typically evident 2–3 days after GnRH agonist administration and can last for approximately 1 week. However, it is subsequently followed by desensitisation of the pituitary-gonadal axis, which suppresses LH, FSH and testosterone levels.10
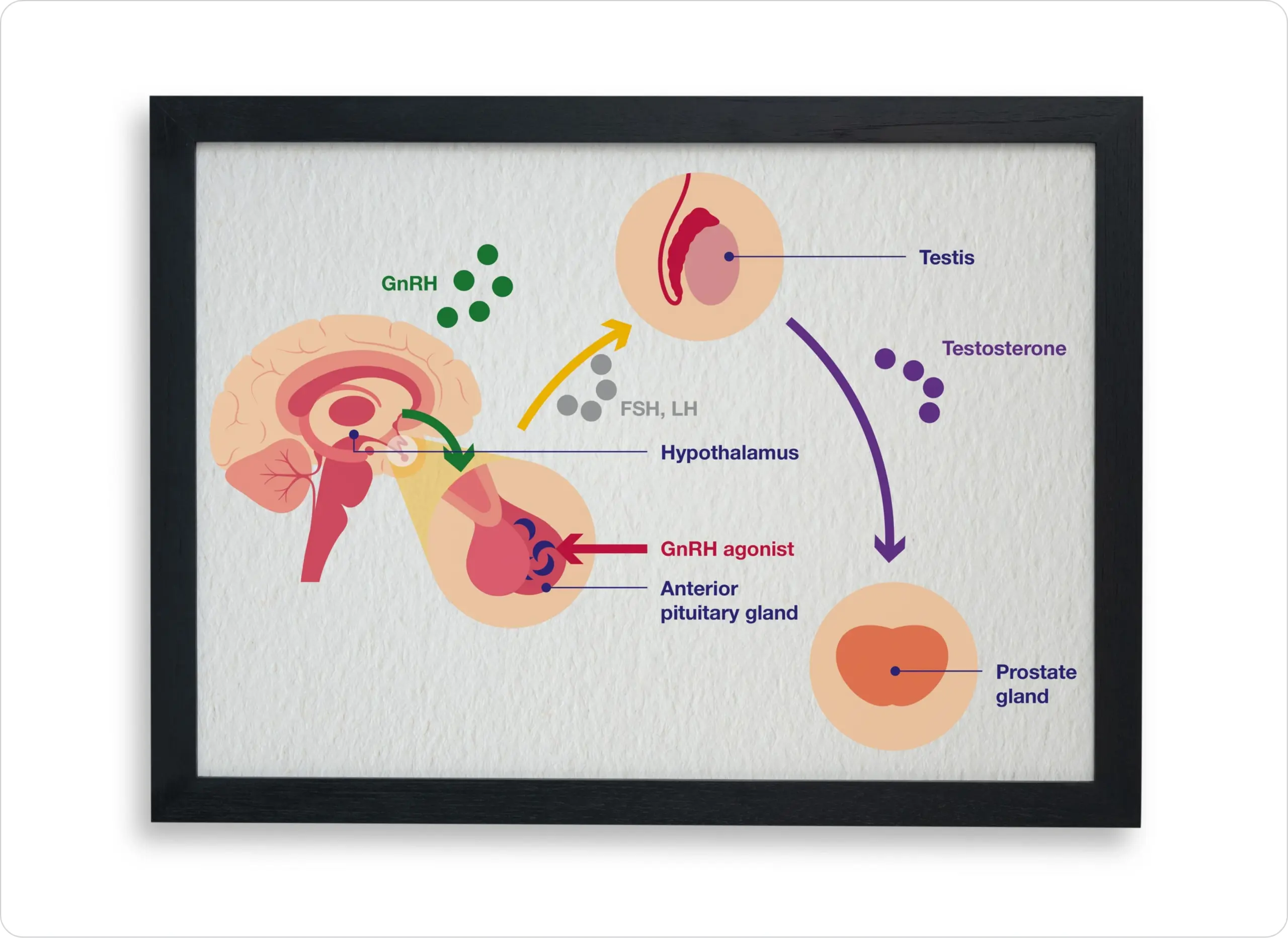
Adapted from Poppel, et al. 2020.10
GnRH antagonists
Available as once-daily oral tablets or once-monthly depot injections, GnRH antagonists act through direct inhibition of GnRH receptors in the anterior pituitary gland, preventing signalling for secretion of LH and FSH, and consequently reducing the production of testosterone.10,12,14
Unlike GnRH agonists, GnRH antagonists are not associated with an initial surge of serum testosterone.10
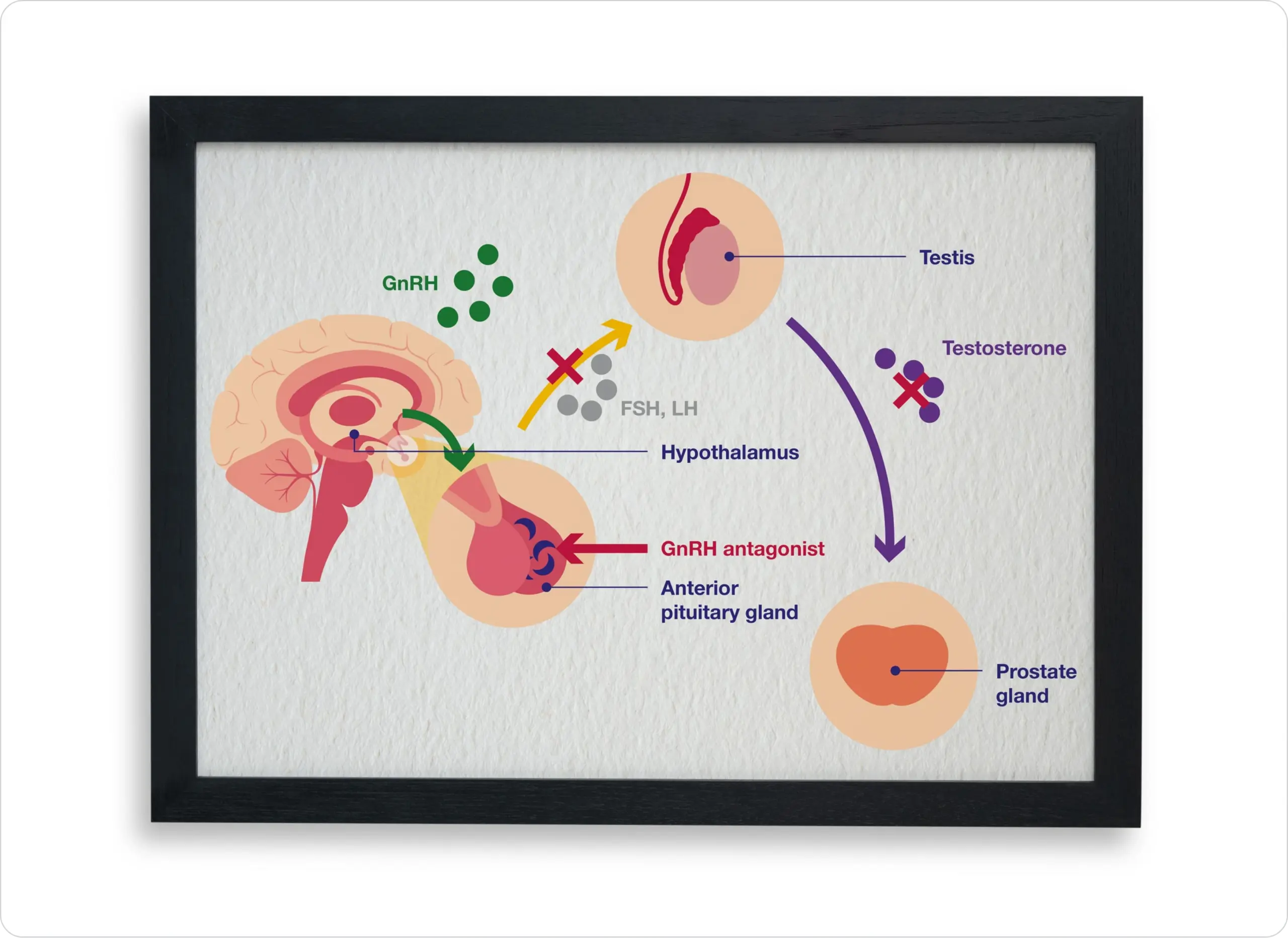
Adapted from Poppel, et al. 2020.10
Androgen receptor pathway inhibitors (ARPIs)
In patients with metastatic castration-resistant prostate cancer, both androgen-dependent and androgen-independent cells contribute to tumour growth. This dual presence may be due to genetic alterations in the androgen receptor, which lead to the proliferation of androgen-independent cells.13
ARPIs work by either inhibiting the enzymes involved in androgen production or blocking the androgen receptor itself.13
Optimising ADT For Advanced Hormone-Sensitive Prostate Cancer
Watch Professor Alison Birtle (Consultant Oncologist – Rosemere Cancer Centre, Lancashire Teaching Hospitals, United Kingdom (UK)) talk through the guidelines for doublet, triplet and monotherapy treatments for advanced hormone-sensitive prostate cancer. This video was recorded in June 2024.
Approx watch time 11 mins

Duration : 10:49 min
Managing Fatigue
Fatigue is a common side effect for patients with prostate cancer undergoing ADT, with as many as 43% of patients reported to experience clinically significant fatigue.15
Fatigue can adversely affect a person’s functional status and quality of life. It may hinder their ability to work, participate in family life and maintain social and personal activities, and can be accompanied by guilt, anger, boredom and loss of self-esteem.16
The following strategies may help you in supporting your patient in managing their fatigue:
![]()
The fatigue diary from Macmillan Cancer Support may help your patients keep track of their daily energy levels.
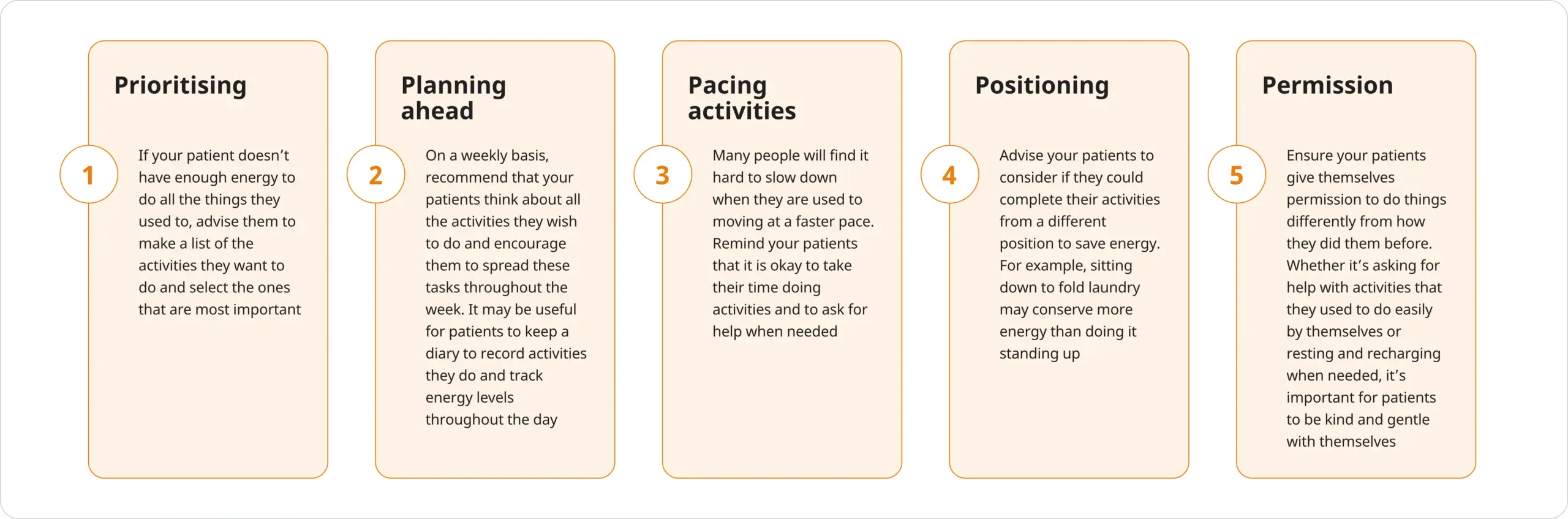
‘The Five Ps’ of fatigue management
You can also advise your patients to use ‘The Five Ps’ to help them make the most of the energy they do have:19
The Link Between ADT And Bone Health
The skeletal system is a common site of prostate cancer metastasis, with the most frequent sites being the vertebral column, pelvic bone, ribs, long bones and the skull. Bone metastases are associated with an increased risk of skeletal-related events, such as fractures and spinal cord compression and surgical or radiation intervention of the bone, necessitating the need for therapeutic or palliative measures.20,21
Although the underlying pathophysiology of bone metastases in patients with prostate cancer remains unclear, high levels of biochemical markers of bone turnover in this cohort are indicative of increased rates of osteoblast and osteoclast activity.20
Following initiation of ADT, serum levels of testosterone and oestrogen decline, disrupting the balance of bone remodelling by stimulating osteoclast activity, decreasing osteoclast apoptosis and increasing osteoblast apoptosis. This results in decreased bone formation and increased bone resorption, leading to a net loss of bone mass.20,22
Fracture risk in patients with prostate cancer receiving ADT
ADT is associated with decreases in bone mineral density (BMD), with studies suggestive of a 5–10% loss of BMD from baseline in the first year after initiating ADT for prostate cancer – greater than the normal age-related bone loss of 0.5–1.0% per annum.22–25
In a study of 50,613 men (≥66 years of age) with a diagnosis of prostate cancer who received ADT (GnRH agonists or orchiectomy), ADT was associated with an increased risk of fracture vs. patients not receiving ADT:21
- Of men surviving at least five years after diagnosis, 19.4% of those who received ADT had a fracture vs. 12.6% of those not receiving ADT (P<0.001)
- In the same four-year period, 5.2% of patients treated with ADT were hospitalised with a fracture, as compared with 2.4% of patients not treated with ADT (P<0.001)
Fracture risk was shown to increase with the number of doses of a GnRH agonist administered during the first year after diagnosis:21
- The relative risk of any fracture was 1.45 (95% CI, 1.36–1.56) among patients receiving 9 or more doses of GnRH agonist in the first 12 months after diagnosis and 1.54 (95% CI, 1.42–1.68) among patients who underwent orchiectomy
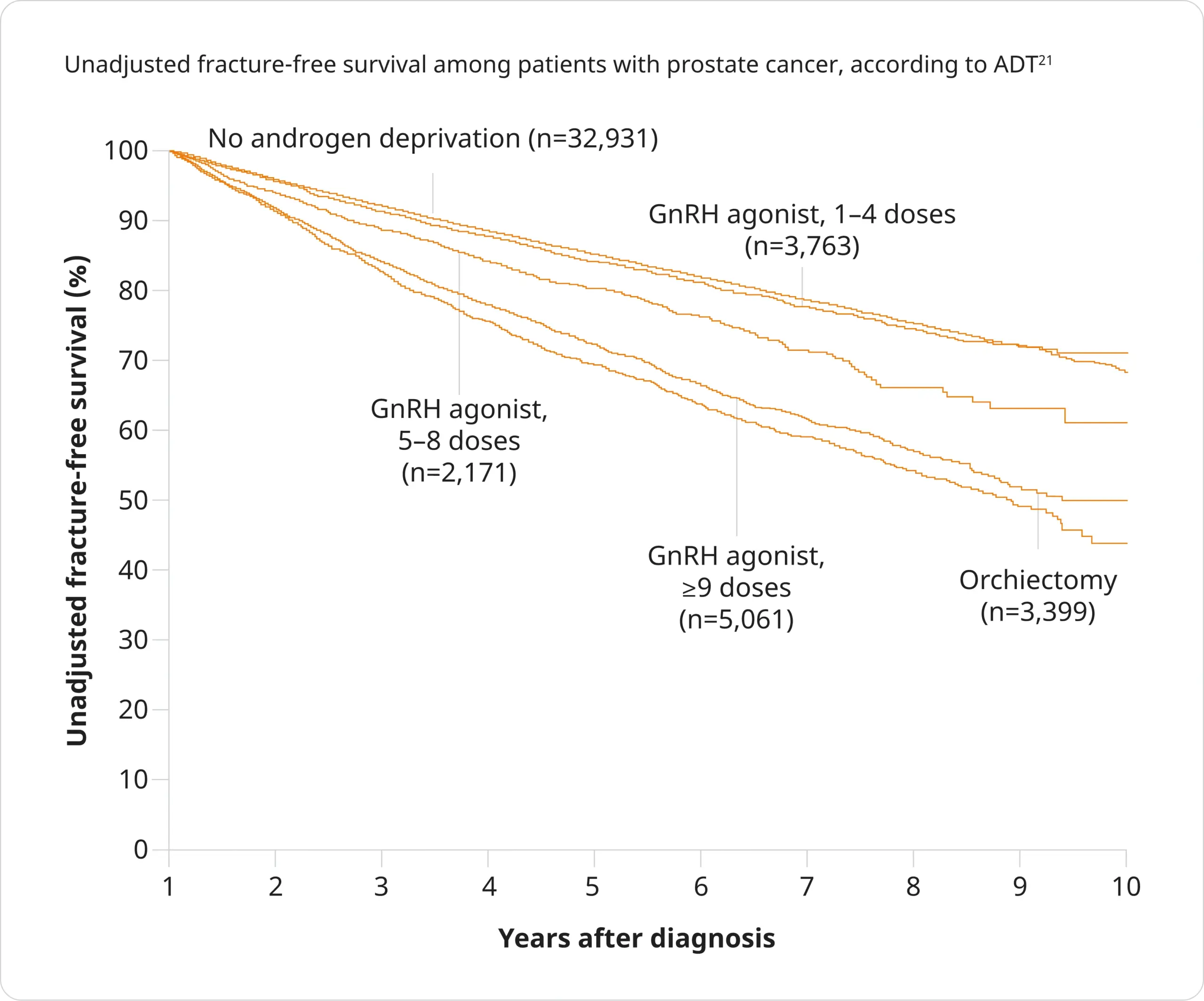
Adapted from Shahinian, et al. 2025.21
Assessing fracture risk in patients with prostate cancer
Risk assessment tools are available to help you determine your patients’ fracture risk:22*
Calculates 10-year probability of fracture, and may be used with or without BMD
FRAX® is based on primary data from 12 prospectively studied population-based international cohorts, comprising more than 60,000 individuals and 5,000 incident fractures, with subsequent external validation in cohorts comprising 230,486 individuals.22,26
The FRAX® algorithms give the 10-year probability of a hip fracture and the 10-year probability of a major osteoporotic fracture (i.e. clinical spine, forearm, hip or shoulder fracture).26
Figure from FRAX® Calculation Tool.27
This tool has not been developed by Accord Ltd, and we are not responsible for the content.
Calculates 1–10 year incidence of fracture
Devised from routinely collected data from general practitioners (GPs) across the UK, the QFracture® calculator works out the risk of developing an osteoporotic (i.e. hip, wrist, shoulder or spine) fracture or hip fracture.28
Figure from QFracture® – 2016 risk calculator.28
This tool has not been developed by Accord Ltd, and we are not responsible for the content.
Managing bone health in patients with prostate cancer receiving ADT
It is recommended that bone health be prioritised when patients with prostate cancer begin long-term ADT.22
Guidance for assessing and managing bone health in patients with prostate cancer has been published in a consensus position statement from key opinion leaders specialising in the management of prostate cancer and bone disorders:22
- All patients starting or continuing long-term ADT should receive individualised, patient-centred lifestyle advice regarding bone health
- Calcium/vitamin D supplementation should be offered if required
- Fracture risk should be calculated using FRAX®, with BMD assessment included where feasible
- BMD should always be assessed where fracture risk calculated using FRAX® alone is close to the intervention threshold
- Intervention should be provided if indicated by local or national guidelines
- Patients requiring bone protection therapy should be further assessed, with referral to specialist centres, if available, and offered appropriate treatment to reduce fracture risk
- Those near to, but below an intervention threshold, and patients going on to additional systemic therapies, should have FRAX® (including BMD) repeated after 12–18 months
The Link Between ADT And Cardiovascular (CV) Health
Cardiovascular disease (CVD) is a leading cause of mortality among patients with prostate cancer, with 27–34% of deaths across all prostate cancer stages attributable to CV causes.29–32
ADT, and specifically GnRH agonists, have been associated with an increased risk of CV events among patients with prostate cancer vs. patients without prostate cancer and vs. patients not receiving ADT.6,33,34
- An observational study of men (aged ≥66 years) with local or regional prostate cancer (N=73,196) found, when compared to men receiving no ADT, GnRH agonists to be associated with an increased incidence of diabetes (aHR, 1.44; p<0.001), coronary heart disease (aHR, 1.16; p<0.001), myocardial infarction (aHR, 1.11; p=0.03) and sudden cardiac death (aHR, 1.16; p=0.004)34
- A prospective Phase 3, randomised controlled trials (n=2,328) showed that, among men with pre-existing CVD, the risk of cardiac events within 1 year of initiating therapy was significantly lower among men treated with a GnRH antagonist compared with GnRH agonists (56% lower risk; HR ratio, 0.44; 95% CI, 0.26–0.74; p=0.002). The absolute risk reduction during the first year was 8.2%35‡
Although the relationship between ADT and adverse CV events remains to be fully elucidated, it is thought that ADT can worsen CV risk factors by inducing metabolic changes, such as dyslipidaemia, dysglycaemia, obesity and hypertension.36,37
Why is it important to assess CV risk in patients with prostate cancer?
Patients with prostate cancer often present with CV risk factors at the time of diagnosis. Hence, it is recommended to account for pre-existing CVD or risk factors before initiating ADT treatment.6,37,38
Hear Dr. Tharshini Ramalingam (Cardio-Oncology Speciality Doctor, Royal Brompton Hospital; GP Partner, Oxted Health Centre; Clinical Champion, Prostate Cancer UK) discuss the importance of assessing CV risk in patients with prostate cancer. This video was recorded in October 2024.
Clinical tools to support assessment of CVD risk in patients with prostate cancer
To support you in practice, the Prostate Cancer Cardiovascular (PCCV) Expert Network have developed clinical tools to prompt estimation of CVD risk and guide management to minimise cumulative CVD risk in prostate cancer patients receiving ADT.6
The following simplified checklist is designed for seamless integration into your practice to support you in assessing CVD risk in your patients prior to initiating ADT.6
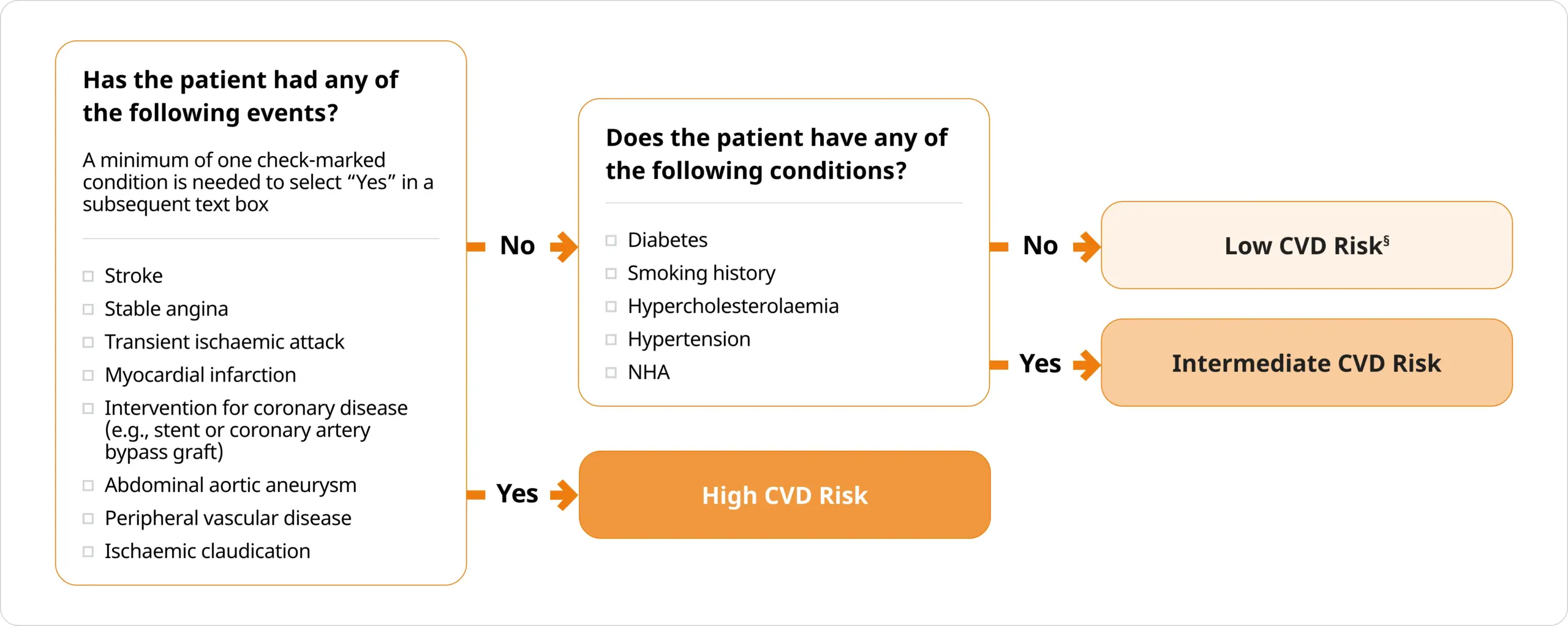
Adapted from Merseburger, et al. 2024.6 Abbreviations can be found in the footnotes of this page.
The PCCV panel emphasised that CVD risk categories are solely intended to guide subsequent management and should not be misconstrued as a predictor of future CV events, unlike the Framingham Risk Score.6
Based on CVD risk categories, the PCCV Expert Network has proposed recommendations for managing and individualising ADT treatment.6

Adapted from Merseburger, et al. 2024.6
Learn more about the PCCV Expert Network recommendations
The “ABCDE” paradigm
Developed by Bhatia et al. (2016) to control CV risk factors in patients with prostate cancer receiving ADT.39
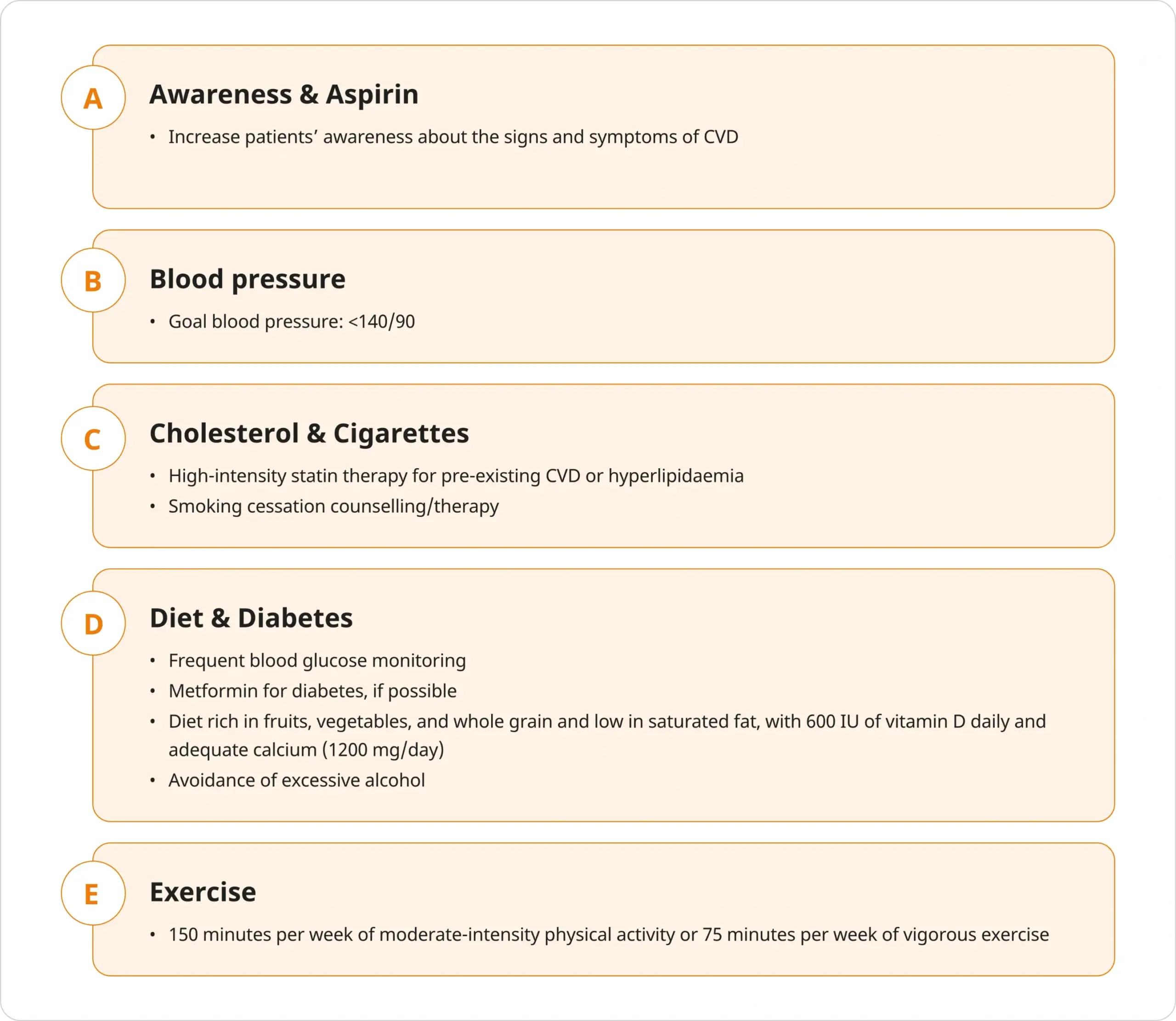
Adapted from Bhatia, et al. 2016.39
Intermittent Therapy
Despite continuous ADT being the mainstay of treatment for advanced prostate cancer, it has been associated with patient morbidity, including sexual dysfunction, hot flushes and loss of BMD.40,41
The initial testosterone surge following initiation of a GnRH agonist can also be deleterious for some patients; such as those with bladder outlet obstruction secondary to locally advanced prostate cancer; those with bone metastases, whereby bone pain could be exacerbated; and patients in whom metastatic disease of the spine threatens to cause spinal cord compression.40
As a clinical strategy, intermittent ADT enables cyclical recovery of serum testosterone levels and has been postulated to minimise adverse events associated with continuous ADT while providing comparable efficacy. Such an approach could be considered for certain patients, including those experiencing side effects from continuous ADT.40–42
Despite formal recommendations from authoritative bodies, such as the National Comprehensive Cancer Network (NCCN) and the National Institute for Health and Care Excellence (NICE), that support the consideration of intermittent ADT, no formal guidelines on the optimal protocol for intermittent ADT have been published.41–43
Patient Perspectives
In the video below, recorded from European Association of Urology (EAU) 2024, Doctor Eva Hellmis, Professor Amit Bahl, Professor Romain Mathieu and Andrew Gabriel talk through:
- Patient perspective of prostate cancer and ADT use (See 01:32)
- An overview of medical and surgical treatments for prostate cancer (See 08:37)
- Investigating the impact of CV disease and bone health in prostate cancer (See 15:18)
Future Direction
Hear Professor Alison Birtle (Consultant Oncologist – Rosemere Cancer Centre, Lancashire Teaching Hospitals, UK) discuss what can be done in the future when treating prostate cancer patients with ADT, and how a better prostate-specific antigen (PSA) response for your patient can lead to better overall survival. This video was recorded in June 2024.
Approx watch time 4 mins

Duration : 04:35 min
Summary of Product Characteristics, legal classification and cost of Accord products:
Abiraterone 500 mg film-coated tablets (Prescription Only Medicine)
Abiraterone 500 mg SmPC – 56 tablets: £2,735 (July 2025)
Degarelix 80 mg, 120 mg powder and solvent for solution for injection (Prescription Only Medicine)
Degarelix 80 mg powder and solvent for solution for injection SmPC: £80.11 (July 2025)
Degarelix 120 mg powder and solvent for solution for injection SmPC: £158.51 (July 2025)
Docetaxel Accord 20 mg/1 ml, 80 mg/4 ml, 160 mg/8 ml concentrate for solution for infusion (Prescription Only Medicine)
Docetaxel 20 mg/1 ml concentrate for solution for infusion SmPC: £145.80 (July 2025)
Docetaxel 80 mg/4 ml concentrate for solution for infusion SmPC: £479.06 (July 2025)
Docetaxel 160 mg/8 ml concentrate for solution for infusion SmPC: £958.11 (July 2025)
Prednisolone 1 mg, 2.5 mg, 5 mg, 10 mg, 20 mg, 25 mg, 30 mg tablets (Prescription Only Medicine)
Prednisolone 1 mg SmPC – 28 tablets: £0.65 (July 2025)
Prednisolone 2.5 mg SmPC – 28 tablets: £3.94 (July 2025)
Prednisolone 5 mg SmPC – 28 tablets: £0.49 (July 2025)
Prednisolone 10 mg SmPC – 28 tablets: £9.70 (July 2025)
Prednisolone 20 mg SmPC – 28 tablets: £19.46 (July 2025)
Prednisolone 25 mg SmPC – 56 tablets: £42.49 (July 2025)
Prednisolone 30 mg SmPC – 28 tablets: £29.12 (July 2025)
Prednisolone 2.5 mg, 5 mg gastro-resistant tablets (Prescription Only Medicine)
Prednisolone 2.5 mg gastro-resistant SmPC – 28 tablets: £1.06 (July 2025)
Prednisolone 5 mg gastro-resistant SmPC – 28 tablets: £1.16 (July 2025)
Reporting forms and information can be found at yellowcard.mhra.gov.uk Adverse events should also be reported to Accord-UK LTD on 01271 385257 or
email medinfo@accord-healthcare.com.
*FRAX® and QFracture® have not been specifically developed for use in patients with prostate cancer.22
†Available in the UK only.26
‡These analyses can only be interpreted as hypothesis-generating.35
§A patient’s risk level may transition from “Low Risk” to “Intermediate Risk” or “High Risk” after 2 or 3 years of hormonal plus NHA treatment.6
¶Referral to cardiologists is recommended, but is subject to each country’s healthcare system and resources.6
ADT, androgen deprivation therapy; aHR, adjusted hazard ratio; ARPI, androgen receptor pathway inhibitor; BMD, bone mineral density; CI, confidence interval; CV, cardiovascular; CVD, cardiovascular disease; EANM, European Association of Nuclear Medicine; EAU, European Association of Urology; ESTRO, European Society for Therapeutic Radiology and Oncology; ESUR, European Society of Urogenital Radiology; FRAX, Fracture Risk Assessment Tool; FSH, follicle-stimulating hormone; GnRH, gonadotropin-releasing hormone; GP, general practitioner; HR, hazard ratio; HSPC, hormone-sensitive prostate cancer; ISUP, International Society of Urological Pathology; IU, international unit; LH, luteinising hormone; NCCN, National Comprehensive Cancer Network; NHA, novel hormone agent; NICE, National Institute for Health and Care Excellence; PCa, prostate cancer; PCCV, Prostate Cancer Cardiovascular; PROSCA, Global Congress on Prostate Cancer; PSA, prostate-specific antigen; RWTH, Rheinisch-Westfälische Technische Hochschule; SIOG, Society of Geriatric Oncology; SmPC, Summary of Product Characteristics; UK, United Kingdom; VACURG, Veterans Administration Cooperative Urologic Research Group.
References:
- Nassar GN & Leslie SW. Physiology, Testosterone. 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK526128/ Accessed: July 2025
- Barone B, et al.Int J Mol Sci. 2022;23(7):3535
- Gurung P, et al.Physiology, Male Reproductive System. 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538429/ Accessed: July 2025
- Corotchi MC, et al.Rom J Morphol Embryol. 2016;57(1):75–80
- Crawford ED. Rev Urol. 2004;6(Suppl 7):S3–S11
- Merseburger AS, et al.World J Urol. 2024;42(1):156
- Lehtonen M and Kellokumpu-Lehtinen PL. SAGE Open Med. 2023;11:20503121231216837
- Drudge-Coates L. Int J Urol Nurs. 2010;3(3):85–92
- Denmeade SR and Isaacs JT. Nat Rev Cancer. 2002;2(5):389–396
- Poppel HV and Abrahamsson PA. Int J Urol. 2020;27(10):830–837
- Choi E, et al.Res Rep Urol. 2022;14:87–108
- Cornford P, et al. EAU – EANM – ESTRO – ESUR – ISUP – SIOG. Guidelines on Prostate Cancer. 2025. Available from: https://uroweb.org/guidelines/prostate-cancer Accessed: July 2025
- Jacob A, et al.Cancers (Basel). 2021;13(21):5417
- ORGOVYX® Summary of Product Characteristics
- Nelson AM, et al.Support Care Cancer. 2016;24(10):4159–4166
- Larkin D, et al.Int J Nurs Pract. 2014;20(5):549–560
- Lopez P, et al.Prostate Cancer Prostatic Dis. 2021;24(2):465–481
- Macmillan Cancer Support. Your fatigue diary. 2023, 9th edition. Available from: https://www.macmillan.org.uk/dfsmedia/1a6f23537f7f4519bb0cf14c45b2a629/13197-10061/mac11664-e09-diary-fatigue-p03-20230914-kp-low-res Accessed: July 2025
- Marie Curie. Managing fatigue. A guide to making the most of your energy. 2022, Edition 4. Available from: https://www.mariecurie.org.uk/globalassets/media/documents/how-we-can-help/booklets-pdfs-only/managing-fatigue.pdf Accessed: July 2025
- Kim TJ and Koo KC. Cancers (Basel). 2020;12(6):1529
- Shahinian VK, et al.N Engl J Med. 2025;352(2):154–164
- Brown JE, et al.J Bone Oncol. 2020:25:100311
- Daniell HW, et al.J Urol. 2000;163(1):181–186
- Berruti A, et al.J Urol. 2002;167(6):2361–2367
- Nguyen PL, et al.Eur Urol. 2014;67(5):825–836
- FRAX®. Available from: https://www.fraxplus.org/ Accessed: July 2025
- FRAX® Calculation Tool. Available from: https://www.fraxplus.org/calculation-tool Accessed: July 2025
- QFracture®. Available from: https://qfracture.org/ Accessed: July 2025
- Shore ND, et al.N Engl J Med. 2020;382(4):2187–2196
- Van Hemelrijck M, et al.BJU Int. 2016;117(3):507–514
- Guo Y, et al.Oxid Med Cell Longev. 2022;2022:8145173
- Weiner AB, et al.Cancer. 2021;127(16):2895–2904
- Li JR, et al.PLoS One. 2022;17(6):e0270292
- Keating NL, et al.J Clin Oncol. 2006;24(27):4448–4456
- Albertsen PC, et al.Eur Urol. 2014;65(3):565–573
- Klimis H, et al.JACC CardioOncol. 2023;5(1):70–81
- Leong DP, et al.JACC CardioOncol. 2024;6(6):835–846
- Lyon AR, et al.Eur Heart J. 2022;43(41):4229–4361
- Bhatia N, et al.Circulation. 2016;133(5):537–541
- Shore ND and Crawford ED. Rev Urol. 2010;12(1):1–11
- Perera M, et al.Nat Rev Urol. 2020;17:469–481
- National Comprehensive Cancer Network. Prostate Cancer. 2025. Available from: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf Accessed: July 2025
- National Institute for Health and Care Excellence. Prostate cancer: diagnosis and management [NG131]. 2021. Available from: https://www.nice.org.uk/guidance/ng131/ Accessed: July 2025





